
- No loading dose required
- Administered over a 60-minute period
- Dosed 10 mg/kg every 24 hours
- For 7 to 14 days in cSSSI
- For 7 to 21 days in HABP/VABP
- Duration of therapy should be guided by the severity and site of the infection, and by the patient’s clinical progress
- Monitoring of serum creatinine is recommended in all patients
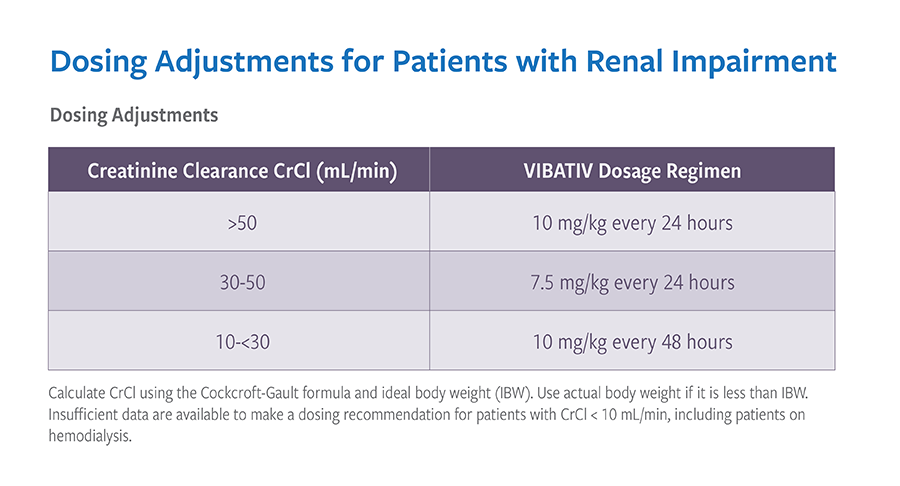
Monitoring Renal Function
- Monitor renal function in all patients as new onset or worsening renal impairment has occurred
- Prior to initiation of treatment
- During treatment (at 48- to 72-hour intervals or more frequently, if clinically indicated)
- At end of treatment
Other Considerations
- No drug interactions were observed
- VIBATIV shares the same potential as vancomycin for infusion reactions if not infused over at least 60 minutes
